Published in October, startling results of natural selection in humans indicates certain immunity genes may have helped people survive the Black Death of the 1346-1350.
Caused by the bacterium Yersinia pestis, bubonic plague has swept across the globe (as pandemics) numerous times over the centuries, resulting in untold numbers of deaths. The most well-known plague pandemic, commonly called the ‘Black Death’, is actually the second, and is estimated to have killed between 30-50% of people across Europe, the Middle East, and North Africa. The first pandemic is known as the Plague of Justinian in 541 AD, estimated to have killed up to 40% of the people in the city of Constantinople. However, it is difficult to estimate how many died during the 6th century plague; this is because there is sparse historic documentation available from the 6th century. What is certain about both pandemics is that each wrought untold amounts of human death that subsequently devastated global economies, empires, and communities. The alarming and devastating mortality rate and the extended length (i.e., years, into decades in many places) of the Black Death (and infectious diseases more generally) can be broadly conceptualized as an evolutionary selective pressure (i.e., something affecting a species or population’s ability to survive and reproduce, known as natural selection).
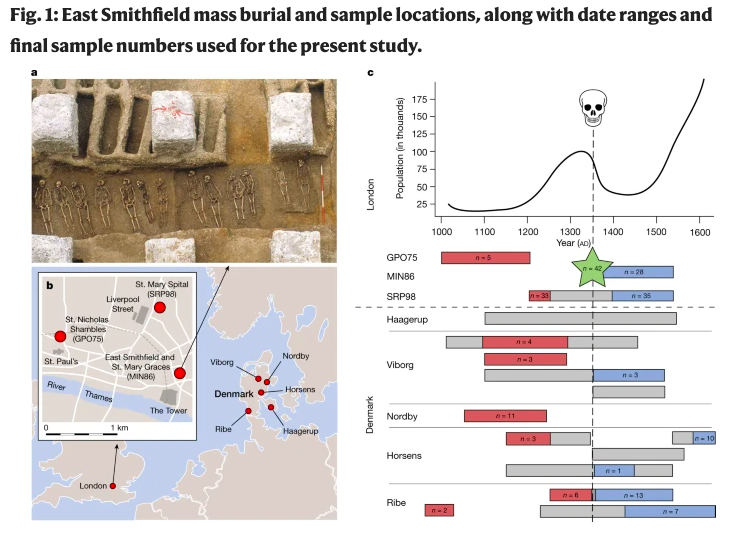
Because of the historic and continuing modern global impact of bubonic plague (and many other infectious diseases) many scientists have tried to directly study Y. pestis by applying ancient DNA methodologies on the remains of plague victims. With heaps of people dying overnight, large communal burial pits (plague pits) were often employed throughout the Black Death due to the inability of communities to bury people individually and quickly enough. However, direct evidence of natural selection in humans and related to the Black Death has been largely elusive because directly linking the genes that helped people survive in the past with the same genes in modern, descendant populations and to Y. pestis has not been found.
The recent study by Klunk et al. (2022) tries to do just that by using an impressive, combined approach with ancient DNA and modern in vetro cell infection experiments. The ancient DNA of individuals who died soon before, during, after the Black Death from London and Copenhagen were analyzed to identify a handful of genes that dramatically changed in frequency. A battery of in vetro experiments were then conducted on cells from modern-day Londoners to test the functional immune responses of the handful of identified genes. One gene variant, rs2549794 of the ERAP2 gene, which helps macrophage cells recognize infectious agents, showed a strong response when exposed to Y. pestis and other pathogens. Specifically, having two copies of the rs2549794 variant may have helped people survive by 40%, compared to having only one copy or none at all. While ERAP2 provides protection against the bubonic plague, it is also associated with increased susceptibility to autoimmune diseases and is a known risk factor for Crohn’s disease.
I enjoyed this article because of the combination, ancient and modern, approach. So often we want to test or corroborate our findings from ancient DNA methodologies using modern techniques and in modern populations. Obviously, many ethical boundaries do not allow for lawful and moral testing of natural selection in humans today. Beyond ethics, the specialized methodologies for in vetro functional analyses or animal testing, as well as the high capital needed to conduct the experiments, limit who and which concepts get tested. Many thought experiments and hypothesis generation takes place among scientists instead (rightfully so). And even with the combination approach used in this recent study, small sample size (number of analyzed individuals) of, both, the ancient and modern populations limits the broader implications of its results. Supplementing sample size with data depth (e.g., expand number of gene analysis, application of various labratory techniques) only helps explain the studied populations; in this case, it is the well-known and extensive historical impact of the Black Death that allows the authors to speculate beyond the studied populations. As the first study to connect Y. pestis to ancient and modern populations’ gene frequencies and functions, more global assessment of plague victims and the ERAP2 gene is necessary to understand the applicability of these results to humans around the world.
Original study: Klunk, J., Vilgalys, T.P., Demeure, C.E. et al. Evolution of immune genes is associated with the Black Death. Nature 611, 312–319 (2022). https://doi.org/10.1038/s41586-022-05349-x
![]()